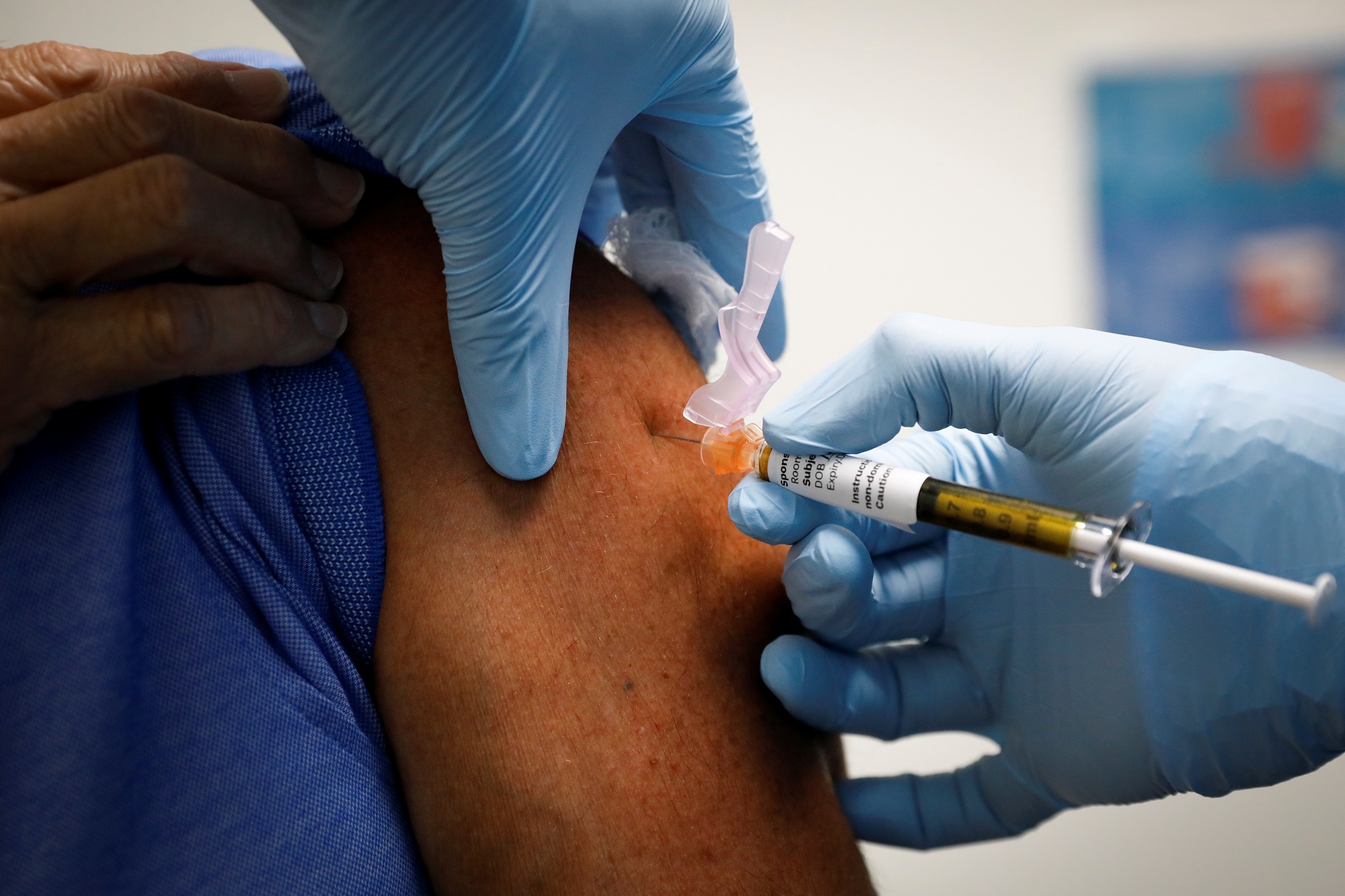
The United Arab Emirates Health Ministry has claimed that the coronavirus vaccine developed by Chinese state-owned pharmaceutical company Sinopharm is 86% effective in fighting against the disease.
The country’s health ministry has been conducting late-stage clinical trials of the vaccine since July to assess its efficacy prior to a rollout. In a statement forwarded by the country’s state news agency on Thursday, the ministry released the results of the trial.
In its tests, the ministry inoculated 31,000 volunteers across 125 nationalities within the UAE. The trials were done while the government released vaccines to around 100,000 people across the Emirates, which the ministry said was part of its voluntary vaccination program.
According to the results, the ministry concluded that the vaccine presented no “serious safety concerns.” However, the report did not say whether any of the volunteers suffered side effects nor did they mention if a placebo was used during the trials.
China currently has five major coronavirus candidates produced by four companies that have reached phase 3 clinical trials. As there are not many positive cases in the country, Chinese companies have conducted tests abroad for the efficacy of their respective candidates.
Countries that had agreed to host the trials were promised early access to the vaccines. So far, the Chinese companies have conducted phase 3 trials in at least 16 foreign countries.
While Sinopharm’s vaccine may not be as effective as those produced by Pfizer and Moderna, the treatment doesn’t need to be stored in sub-zero temperatures. This makes the transport and distribution of the Chinese vaccines easier, particularly in countries that lack cold storage capacities.
Prizer and Moderna’s vaccine uses messenger RNA (mRNA) to stimulate an immune response. The technology is relatively new and has never been used in existing vaccines. The only downside to the technology is that vaccines need to be stored in specialized containers at temperatures of -20°C for Moderna and -75°C for Pfizer.
Meanwhile, Sinopharm is using a tried and tested approach used in vaccines for polio and the flu. This type of vaccine can be stored in normal refrigerators in temperatures of between 2˚C to 8˚C.